General microscopy
 For general microscope work, a magnification between 40X and 400X is usually sufficient and will allow for relatively easy identification of most common ectoparasites.
For general microscope work, a magnification between 40X and 400X is usually sufficient and will allow for relatively easy identification of most common ectoparasites.
Higher magnification
When talking about higher magnification it is important to understand the difference between simply magnifying the size and increasing the amount of visible detail. With high-power microscopy the most important consideration is the image resolution, which means being able to distinguish two objects that are close together as being separate, distinct entities.
For technical reasons there is a limit to the amount of resolution that can be obtained with an ordinary light microscope. Usually, beyond 1000X magnification there is no significant increase in resolution, and any extra magnification merely gives an increase in size without any extra detail.
As you might guess, increasing resolution comes at a price – and means investing in more expensive components such as oil-immersion objectives and condensers. For example, a fairly good oil-immersion objective lens can cost over £300! It is possible to increase resolution beyond 1000x, but the type of equipment required is prohibitively expensive.
The reasons for using oil-immersion objectives are technical – but basically the oil reduces light being reflected back from the slide as well as bending the light rays inwards to focus on the specimen – giving more light, less interference and therefore better detail
Using an oil-immersion objective
- Focus on the specimen using a dry objective lens (40x), making sure that the specimen is central in the field of view.
- Move the dry objective to one side and place a drop of oil on top of the specimen. Swing the oil-immersion lens into place, ensuring that it’s front lens makes contact with the oil drop. If it doesn’t, slowly lower the objective while watching from the side until it just touches the drop
- If you are using modern parfocal lens, which means that all objectives are approximately in focus at the same setting, the specimen should already be roughly in position. Look down the microscope and make any necessary fine adjustments to the focus, iris diaphragm and condenser to obtain the best image. Be very careful not to lower the objective onto the slide as the working distance with high magnification lens is very narrow.
- After use it is important to clean the oil from the lens using a soft tissue. It should be cleaned using a wiping movement, rather that a circular, scrubbing movement as this might scratch the lens. Any dried on oil can be removed using xylene, but be careful as this could dissolve the cement securing the lens!
So what can you see at 1000x mag?
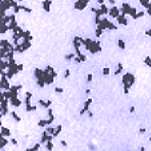
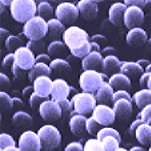
You will see the internal structures of algae and parasites – if you can get them to stay still! However, at this magnification we would normally be looking at bacteria and other micro-organisms. In honesty, you may be initially disappointed with what you actually see. Most micro-organisms are transparent and even at this magnification are difficult to see with an ordinary bright-field microscope. With special stains they are visible, but still fairly small and insignificant (see photos).
 |
 |
|
Gram stained bacteria as viewed with a light microscope |
The same bacteria viewed using a scanning microscope |
However, as understanding develops it is possible to determine significant information and interest from high-power microscopy. Special staining techniques such as Gram and acid-fast staining enable us to determine the basic group of bacteria – for example most bacterial fish pathogens are Gram-negative and stain red with Gram staining.
It is also possible to determine approximate size and thereby differentiate between long Flexibacter. sp and the smaller ulcer-related bacteria such as Aeromonas sp. The shape of the bacterium can also be seen as either straight rods, bent rods or round (cocci).
While conclusive identification can only be determined by culturing the bugs and subjecting them to biochemical testing, high powered microscopy enables us to go part way to discovery this normally hidden, fascinating world
.
